Learn the science behind:
Impact of Vinegar & Pineapple on The Texture of Fried Eggs
If you google vinegar + eggs, you’ll find plenty of articles discussing: “why to use vinegar when poaching eggs” OR “how to dissolve eggshells with vinegar”. But none explaining why vinegar added to raw egg whites changes the texture of our fried egg. And that’s what we wanted to know!
So, we decided to figure it out ourselves. We think we’ve found why it’s not discussed frequently. Apparently, the effects are mostly pretty undesirable…
The egg frying experiment
Our journey started with a question from one of our readers, Carolyn. She wanted to know why adding vinegar or pineapple juice to an egg white changed the texture of a cooked egg.
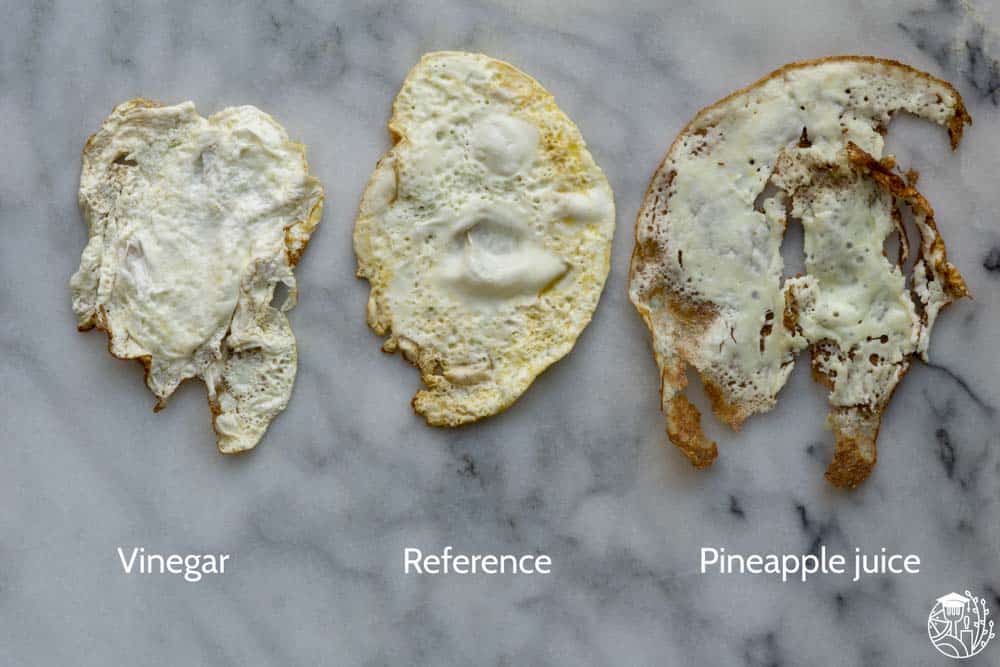
So, we got three eggs. We split the whites from the yolks. We put the yolks aside for another application. One egg white we kept as is. To the others, we added either a teaspoon of vinegar or a teaspoon of the juice of a freshly cut pineapple.
After leaving them for 15 minutes or so, we fried our egg whites and had a taste. And, just like Carolyn, we noticed some big differences. The vinegar sample tasted watery. The pineapple one was very thin and clearly tasted different.

We need to start with the proteins
So, what’s going on? Google didn’t have a clear-cut answer, so we set out to investigate. When studying egg whites, you’ll almost always need to study their proteins. About 10% of egg whites is made up of protein. The remainder is mostly water, and a few minor components.
Want a quick refresher on the science of eggs? Have a look at our infographic.
Proteins are long strands of amino acids. These long linear strands curl up in complex 3D shapes. These 3D configurations are very important for proteins. It’s what makes them work. When they lose this configuration, their properties change.
Frying eggs denatures the proteins
When you fry an egg, you will notice two main processes:
- The egg turns white, whereas it started out translucent
- The egg turns solid, whereas it was liquid to start with
Both these processes are due to changes in the protein structure. When heating an egg, those complex 3D structures unravel, causing the egg white to change. This process is called protein denaturation.
Denatured egg whites will start to bond with each other and form a gel. This transformation from a liquid into a solid-like consistency is called coagulation. The gel is made up of strands of unfolded proteins that hold onto water. This is what gives the fried egg whites their characteristic texture.
Need a proper introduction to proteins? Our Food Chemistry Basics course takes a deep dive into proteins!

Syneresis aka: Weeping eggs
A perfectly fried egg forms a gel in which egg white proteins hold onto moisture. However, if you treat the proteins a little too harshly, the eggs might start to weep. This is especially common when making scrambled eggs.
Weeping is caused by a process called syneresis. It is often caused by too much intense heat. As a result, the proteins pull even tighter in the gel. Instead of holding on to that moisture, it now pushes it out.
Acids can also change proteins
Heat is a very common reason why proteins denature. However, there are several more. For our egg whites specifically, acids are important as well.
Vinegar is an acid. Acids are ingredients that have a pH value below 7. The lower the value, the more acidic an ingredient is, down to a value of 0. Vinegar is quite acidic, with a pH value of 2-3.
Just like heat, acids can change the 3D configuration of proteins. They do this by modifying the interaction of amino acids with one another. Amino acids in the protein can interact with one another. Some may be attracted to one another, whereas others are repelled. This interaction is crucial for determining how a protein folds and forms its 3D structure.
Under acidic conditions, the interactions between amino acids are modified, causing the protein to change and denature.
Technically, you can ‘cook’ proteins with acids. Just like you could use heat. You use this exact technique when making ceviche – an acid ‘cooked’ fish.
Cutting up proteins
Proteins can lose their 3D structure due to denaturation. However, this is not the only way proteins can change. Under certain conditions, the long strand of amino acids can also be broken down into smaller pieces. This drastically changes its properties. One effect is that shorter chains aren’t as good at forming gels as the larger chains are. For a gel to form, the strands need to hold onto water, and that is harder to do for these shorter strands.
Pineapple juices ‘eat’ proteins
One way to break down proteins is by using protein-digesting enzymes, called proteases. These highly specialized molecules – which also happen to be proteins – can cut proteins into smaller pieces.
Pineapples happen to contain these proteases, called bromelain. As such, pineapple juice added to an egg white can cause the proteins to break down into smaller pieces if given enough time. The proteases in pineapple juice are sensitive to heat. So once the juices have been heated, they loose this ‘cutting’ tool.
The proteases in pineapple can be beneficial in some cases, but cause problems in others. For instance, it can be used favorably as a meat tenderizer. However, it can also break down a gelatin gel.
Evaluating our fried eggs
So let’s have a look at our fried eggs again. First up: the vinegar sample.
Vinegar causes fried eggs to weep
Remember how our vinegar eggs were watery? When taking a bite and lightly chewing the egg, it seemed as if a lot of water was released. It made for an unappetizing experience for sure.
Knowing what we now know about egg white proteins the vinegar must have started to denature the egg whites before cooking. It’s also why the vinegar + egg white sample turned a little cloudy and white before frying.
Because the vinegar had already started to cook, we think the additional heat from frying the egg caused ‘syneresis’. The proteins were treated too harshly, causing them to coagulate so much that they started to expel water.
Pineapple juice makes a thin egg
When frying the pineapple juice egg white, we immediately noticed that the egg was a lot thinner and had a significantly larger surface area. It also tasted a little mushy. We expect this is due to the pineapple protease. This native pineapple enzyme will have broken down part of the egg white proteins. This thins out the egg white. The smaller pieces can’t hold onto each other as well.
Upon eating the pineapple egg white had a strange off-flavor. Again, this is likely caused by the enzymes. Adding pineapple to milk products is known to result in the formation of bitter off-flavors. Likely something similar happened to our egg white proteins.
Improving your eggs using acids
When making fried eggs, the addition of vinegar and pineapple juice was not a resounding success. Whereas we can’t come up with a way to use pineapple juice beneficially in your eggs, you can use acids to make better eggs!
Scrambled eggs
Adding acids will change a cooked egg’s texture. Some use this when making scrambled eggs. Adding a little vinegar just before scrambling, makes a lighter texture.
Foams
You can also add acids when making an egg white foam! Again, the acid will denature the proteins slightly. This helps with whipping up a foam.
Poaching eggs
The same phenomenon, which is described countless times online, is used when making poached eggs. By adding vinegar to the boiling water, you help the egg proteins to set. That way, they set a little more quickly, keeping the poached egg more compact.
It shows that knowing how these ‘tricks’ work, helps you to apply that same knowledge to something else. So even if Google doesn’t have your immediate answer, we can decipher it from other applications!
References
Nicholas Clee, Scrambled eggs with vinegar, Sep-11, 2009, link
IFST, Protein: coagulation, link
Johny, L.C., Kudre, T.G. & Suresh, P.V. Production of egg white hydrolysate by digestion with pineapple bromelain: optimization, evaluation, and antioxidant activity study. J Food Sci Technol (2021). https://doi.org/10.1007/s13197-021-05188-0 – Demonstrates that pineapple juices can break down egg white proteins
Harold McGee, On Food and Cooking, 2004, Scribner, p. 384, 808
What's your challenge?
Struggling with your food product or production process? Not sure where to start and what to do? Or are you struggling to find and maintain the right expertise and knowledge in your food business?
That's where I might be able to help. Fill out a quick form to request a 30 minute discovery call so we can discuss your challenges. By the end, you'll know if, and how I might be able to help.






