Learn the science behind:

How to Draw Molecules & Structural Formulas for Food Components
Imagine having the pages of a book, but having no clue in which order they should go. Or having a recipe with only an ingredient list, no instructions on how to make the food. Or having an IKEA cupboard, with all the individual pieces, but no manual. All of this is pretty similar to knowing which atoms a molecule is made up of, but not knowing how to sit attached to one another. If you don’t know that order of pages, those recipe instructions or that manual, it is hard (not always impossible) to figure out how the final thing should look like.
Good thing we came up with page numbers, recipe instructions and a manual. And luckily chemists came up with structural formulas. These formulas tell us how a molecule is build up, how atoms are connected and thus how a molecule truly looks like. In this post we’ll introduce you to these structural formulas and of course, we’ll be applying them for molecules in food.
Molecular formulas
In a previous post we’ve introduced the concept of molecules and atoms to you. We learned that there are a limit number of atom types (also called elements) and that these different atoms can form molecules, such as fats, carbohydrates and proteins. In a next post we discussed how chemists use abbreviations to explain which atoms are present in a molecule, the molecular formulas. An example of such a molecular formula is H2O. This formula describes water which is a molecule made up of 2 hydrogen atoms and 1 oxygen atom.
As we described in the introduction, molecular formulas are like a recipe without instructions, we know which ingredients we need (we have a list of atoms), but we don’t know how to connect these.
Structural formulas
That’s where structural formulas come into play! These formulas are slightly more complicated but tell you exactly how the atoms in a molecule are connected to one another. Let’s demonstrate this using some examples. The molecule you see below is methane, it’s not a molecule common in food, but it’s a good example to start with. The molecular formula of methane is CH4. It shows that methane has a carbon (C) atom in the center and 4 hydrogen (H) atoms attached to the center. As you can see, we are using the same abbreviations for the atoms in structural as well as molecular formula.

Another simple example is shown below. It shows two molecules with the same chemical formula: C4H10. However, their structural formulas are quite different. Again, the formulas clearly show how the atoms are connected.

These were simple structures, with just two types of atoms, let’s discuss one more which is a little more complex. Below you can see the structural formula of the most simple amino acids, alanine. You can see four different atoms in this molecule (N = nitrogen, O = oxygen). You can also see that there is a double line between one O and C atom. This is a double bond.

Simplifying structural formulas
Chemists are lazy, at least, they are lazy when it comes to drawing structural formulas. In real life you won’t often see alanine depicted the way we showed it just now. Instead, chemists will leave out certain atoms and also group atoms into very common structural combinations.
Leaving out hydrogen & carbon atoms
In most drawings hydrogen atoms attached to a carbon atom will not be shown. These hydrogen atoms don’t tend to be very important for chemical reactions. Also, there tend to be a lot of them (as you saw in the examples above).
These hydrogen atoms are left out completely, you won’t see them in the formulas anymore. But also the carbon atoms to which they’re attached will have disappeared. Since carbon atoms also don’t tend to play very important roles in chemical reactions and since there are often just too many, they will also be left out. However, instead of leaving them out completely (letter + connecting line), you will still see the lines, however, the letter C is omitted

Grouping common sets of atoms
In the simplified formula of alanine you can also see that not only the H & C places have been simplified. Also the OH and NH2-groups have been simplified. Instead of drawing the lines, the lines have been left out, since it would else become too messy. This grouping of atoms is very common for groups with hydrogen atoms and one other type. You might be asking why these hydrogen atoms aren’t left out completely either. That has to do with somewhat more complex chemistry. These hydrogen atoms play more important roles in chemical reactions. A chemist doesn’t automatically know how many there are, so it has to be shown.
Rules for building molecules
That brings us to the ‘rules’ for building molecules. When forming molecules atoms use their electrons to form bonds with one another. The number of atoms available for such bonding determines with how many other atoms an atoms can bind. By knowing this, chemists can understand how atoms will come together.
As you have seen in the previous examples, carbon (C) has the ability to form four bonds, oxygen (O) only two, nitrogen (N) three and hydrogen (H) only 1. Since chemists know this, they will still know how many atoms there are in a structural formula, even if some have been left out.
Showing 3D structures
For a lot of molecules in food, these simple skeletal versions aren’t enough to show a molecule’s real structure, or they are slightly inconvenient. This occurs when it’s important to know whether a certain atom sits at the front or back of a molecule. Or when the structure has a loop inside. For these purposes slightly more advanced techniques have been developed.
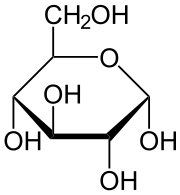
One of these is shown below. This glucose molecule has a circle in the structure. You can see that the front of a circle is slightly thicker than the back. This way of drawing molecules is one that is commonly used for carbohydrates since it helps to see the 3D structure.
In the structure below you can see another method to indicate the 3D structure of a molecule. The dotted lines in this structure of vitamin B12 indicates that the group of atoms points backwards. The thicker triangle bond shows that groups point forward. You might be wondering why this is important at all. Well, in chemistry, the right groups have to be together to interact and react. If you know your reacting group sits at the front, but also know the molecule it can react with, doesn’t have a place to see because of a lot of other groups being in the way, you reaction most likely won’t proceed so easily anymore.

Limitations of structural formulas
In food a lot of molecules are too large and too complex to be shown in structural formulas. A common example are proteins. Proteins tend to be enormous molecules and drawing them in such detail would be far to complicated and wouldn’t necessarily give you a lot of information.
That said, in a lot of other applications structural formulas are very convenient, as we show for instance when discussing lard vs. olive oil. You will see structural formulas being used in a lot of other areas on this blog as well (e.g. when discussing vitamin C oxidation or the Maillard reaction).
What's your challenge?
Struggling with your food product or production process? Not sure where to start and what to do? Or are you struggling to find and maintain the right expertise and knowledge in your food business?
That's where I might be able to help. Fill out a quick form to request a 30 minute discovery call so we can discuss your challenges. By the end, you'll know if, and how I might be able to help.






You made such an interesting piece to read, giving every subject enlightenment for us to gain knowledge. Thanks for sharing the such information with us to read this… food14